Stop or I’ll Soot!!!
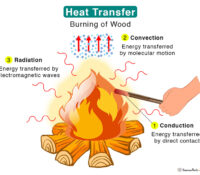
Fire. Something about fire touches our brainstems…both good and bad! Uncontrolled fire is terrifying and deadly to be sure. But the controlled burning of wood at a campfire or in a fireplace in your home almost can’t be beat, to my mind! For that very reason, a fairly common amenity to houses nowadays is the gas log fireplace insert.
When not installed properly, these logs will generate soot. These soot particles can leave the fireplace and meander. All. Over. Your. House. Read More