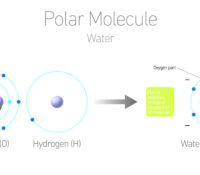
One of the Most Amazing Compounds Around – Water?!?!
Around Christmas day of 2022, a mass of very cold air settled in the deep south/southeastern United States (where I live). It remained below freezing for over 24 hours. That is very unusual down here. There was a spate of burst pipes all over the area. Why? Water is one of a very few compounds that expands when it freezes…by approximately 9%!! If a volume of water is sealed off in a section of piping, and that water freezes, it is likely the pipe will rupture. Then, when it warms up again and thaws, liquid water will leave the pipe for an adventure!! Read More